  
Therefore, from this concept we can write: 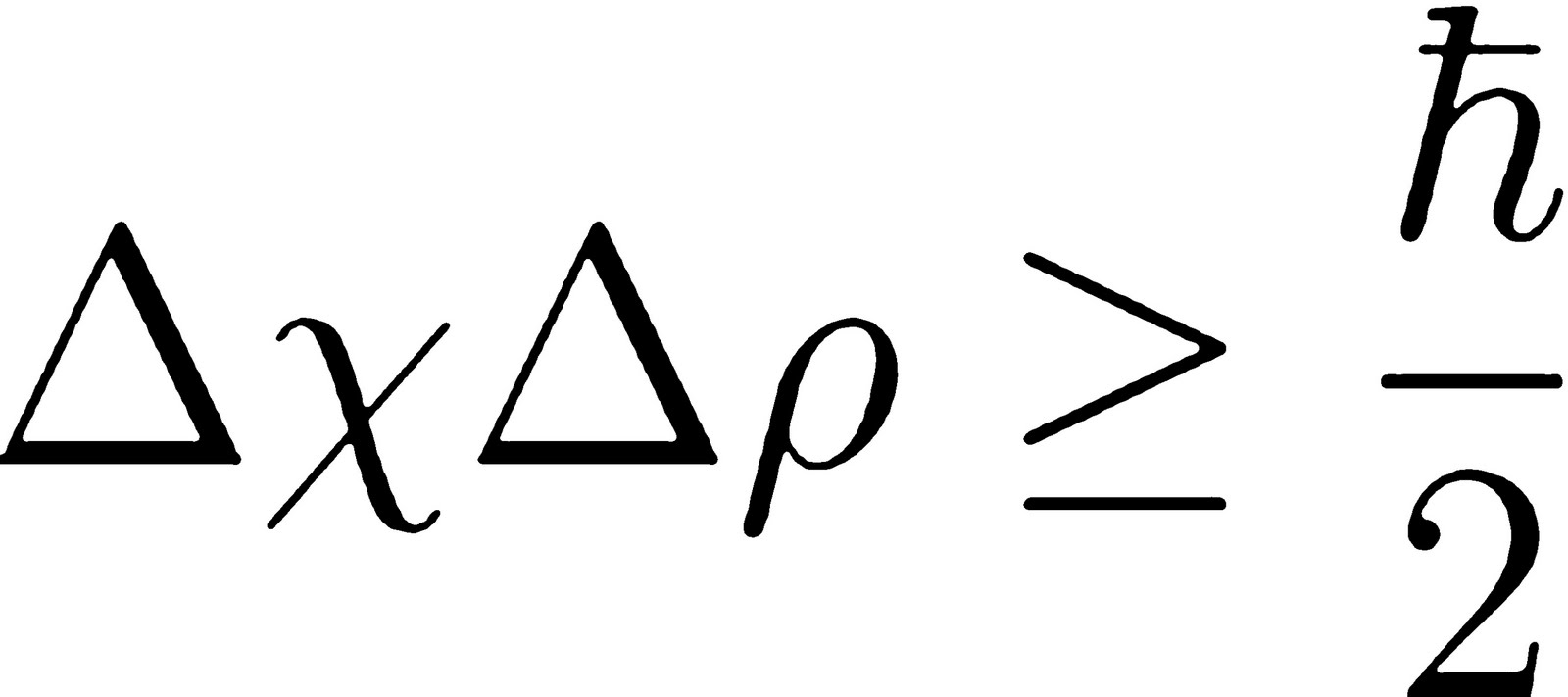
In this principle, ∆x is considered to be an error in the position measurement whereas ∆p is the error in momentum. So, an accurate measurement of one quantity leads to large uncertainty in the measurement of the other.ĭerivation of the Heisenberg Uncertainty Principle Whereas the particle having a well-defined wavelength gives the precise velocity. This shows that particles which have definite position have no certain or fixed velocity. The more the undulation of the particle, the more ill-defined becomes the wavelength, which helps in the determination of momentum of the particle. As each particle has a wave nature, the probability of finding particles is maximum where the undulations of the wave are greatest. 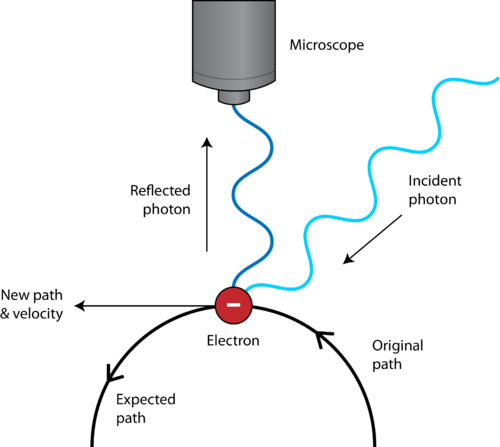
The main reason behind the origin of the uncertainty principle is the dual nature of a wave-particle. Here ∆ denotes the uncertainty in that variable and h is the Planck's constant and its value is 6.6 × 10 −34 joule-second. Heisenberg principle can be numerically written as: ∆X × ∆p ≥ h/4π From this concept it is clear that uncertainties in the conjugate pairs of momentum/position and energy/time were termed by Heisenberg as having a minimum value corresponding to the Planck’s constant divided by 4π. The same relationship can also be applicable for energy and time, as we cannot measure the exact energy in a given finite amount of time. This principle works in contrast to classical Newtonian physics which states all variables of particles to be measurable to an arbitrary uncertainty given good enough equipment.Īs per the uncertainty principle if you know the exact position of a particle it is impossible to determine the exact momentum of the particle and vice versa. However, this principle mainly focuses on position and momentum of particles, the principle states that the more precisely the position is known the more uncertain the momentum is and vice versa. This principle states that there is inherent uncertainty in the act of measuring a different given variable of particles. This principle is one of the most celebrated results of quantum mechanics and it states that we can determine different things about particles simultaneously at a time. This principle was stated by the German physicist Werner Heisenberg in the year 1927. The mountain does not appear to be going anywhere, and neither is the bullet.The uncertainty principle is called the Heisenberg uncertainty principle. If you are measuring the position and momentum of a bullet that is stuck in a cliff of a great mountain somewhere, it is a simple matter. However, in reality you have to do two things to get your answer. You might imagine that it would be easy to get both the exact position of something and its exact mass, path, and speed at the same time. The math is a way of describing things that happen in the real world. That dim spot of light represents the photon or other atomic particle which tunnels through the wall. p115 In the animation to the right, you can see a faint white puff on the right side of the wall after a big puff hits the wall from the left. However, electrons can move through solid walls. In our everyday life, people cannot move through walls. p114 Another interesting uncertainty phenomenon, which makes possible many electronic devices, is called quantum tunneling. American physicist Brian Greene gave the analogy of a moth which flies placidly around in a large closet but who flies frantically back and forth and up and down when placed in a glass jar. However, if the box were made smaller then we would have a more certain idea of where it was located, and because of that we would have to have a less certain idea of how it was traveling. Heisenberg's idea is that if something like an electron were shot into a big box with a certain speed and in a certain direction, then it would be possible to calculate a fairly accurate idea of what its path would be like in the future. However, many other scientists disagree with Einstein. That is, he thought that there is no "uncertainty" in nature, and that the uncertainty exists only in our knowledge about it. p99 He thought that Heisenberg's discovery showed that human knowledge is limited, but he also thought that nature is absolute. p96Īlbert Einstein thought that such a quantum theory could give us only a partial description of nature. Certain pairs of measurements such as (a) where a particle is and (b) where it is going (its position and momentum) cannot be precisely pinned down. It relates to measurements of sub-atomic particles. Heisenberg's uncertainty principle is one of the most important results of twentieth century physics. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
